The Food and Drug Administration’s (FDA) authorization to divide Jynneos vaccine doses into fifths in order to expand the available supply has created new obstacles for the state and local health authorities in their efforts to respond to monkeypox.
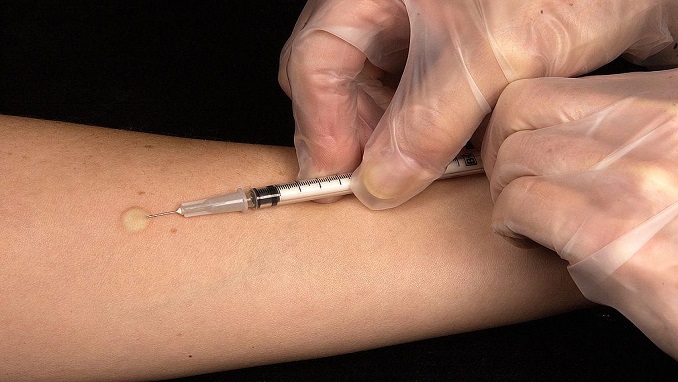
The new method of administering the Jynneos smallpox vaccine the FDA authorized last week, involves injecting one-fifth of the normal dose of the jab – which is now being used to prevent monkeypox amid the ongoing outbreak- intradermally, between layers of skin, instead of into the muscle as most vaccines are administered.
The method is recommended to extend the current supply of doses in the US, but it’s also based on a 2015 study that found administering a reduced dosage of Jynneos intradermally produces a comparable immune response to the one produced by a full dose administered intramuscularly.
The FDA considered authorizing the method, which would expand the supply of Jynneos doses fivefold, after it had become clear that the current supply of the vaccine would not meet the current demand.
Many state and local health authorities in the areas most affected by the monkeypox outbreak are unsure about how to immediately adopt the new strategy after facing several obstacles in going ahead with the federal government’s new guidance.
For example, they don’t have trained workers to administer shots intradermally, they’ve faced a lack of specific needles used for this type of administering the shots as well as a shortage of funds for more supplies.
Intradermal shots are not unheard of within health care although they’re used less frequently than shots administered into the muscle. Both tuberculosis and hepatitis B vaccines are administered by creating a “pocket” between the epidermis and the dermis.
However, some public health workers and private providers who may not be as familiar with the injection method would have to be given some training.
Apart from the costs and logistics aside, the FDA’s guidance also created some uncertainty prompting Paul Chaplin, the CEO of Bavarian Nordic which manufactures Jynneos, express reservations due to the very limited safety data available.

Be the first to comment